
Medicines play a vital role in human lives, they help fight diseases, recover faster, manage chronic conditions, and maintain overall health. People everywhere, including in the UK, expect the medicines they take to be safe, effective, and of the highest quality. Even a single mistake can put lives at risk, which is why it is the prime responsibility of manufacturers to ensure there is no negligence at any stage of production.
To meet this responsibility, manufacturers follow strict guidelines and systems designed to make sure every medicine is produced correctly and consistently. This is where Good Manufacturing Practice (GMP) comes in. GMP ensures that every medicine is made consistently and correctly, giving patients confidence that their treatments are safe and reliable. UK pharmaceutical manufacturers must comply with GMP guidelines enforced by regulatory authorities such as the MHRA, EU, UK Pharmaceutical regulations etc.
In this blog, we’ll explore how Pharma ERP Software in the UK supports GMP compliance and helps manufacturers stay audit-ready.
What is GMP Compliance?
Good Manufacturing Practice (GMP) is a set of rules that ensures medicines are made safely, consistently, and of high quality. It covers every step of production, from raw materials to the final product, so patients or consumers can trust the medicines they use.
GMP isn’t just about rules; it’s about making sure medicines are safe and effective for everyone. Simply put, GMP ensures that every medicine reaching patients is safe, reliable, and effective.

Examples of GMP Compliance in Pharma:
- Clean and controlled production areas to prevent contamination
- Recording every step of production for traceability
- Checking and testing products at every stage
- Making sure machines and equipment work properly
- Training staff to follow standard procedures
- Storing materials and medicines correctly to keep them safe
How GMP Compliance Was Managed Earlier & Its Shortfalls
GMP compliance in pharmaceutical companies was mostly managed through manual processes and paper-based documentation. This typically included:
- Paper-based batch records
- Physical documentation and registers for production and quality activities
- Manual quality checks and approvals
- Separate systems for production, quality, and inventory management
While these methods served their purpose for many years, they also created several operational challenges. Managing large volumes of documents was often time-consuming, and retrieving specific records during regulatory audits could be difficult. In addition, manual systems increased the chances of human errors, missing information, and delays in reporting.
Some common challenges of this traditional approach included:
- Large volumes of paperwork – Maintaining batch records, quality logs, and compliance documents manually required significant time and effort.
- Difficulty in retrieving information – Locating specific records during regulatory inspections could take hours or even days.
- Higher chances of human error – Manual data entry increased the risk of incorrect or incomplete records.
- Limited real-time visibility – Production and quality teams had limited ability to monitor issues or deviations as they occurred.
- Slow audit preparation – Collecting and organising documents for regulatory inspections often became a stressful and lengthy process.
As pharmaceutical operations expanded and regulatory expectations became more stringent, relying solely on traditional systems gradually became less efficient and more risky for maintaining GMP compliance.
What Auditors Look for in GMP Compliance in the UK Pharmaceutical Industry
Regulatory audits are a critical component of the pharmaceutical industry. In the UK, regulatory bodies conduct inspections to ensure that pharmaceutical manufacturers comply with Good Manufacturing Practice (GMP) guidelines and maintain strict quality standards throughout the drug manufacturing lifecycle.
These inspections are designed to verify that medicines are consistently produced, controlled, and documented according to approved procedures. The goal is to ensure that pharmaceutical companies have robust quality management systems, validated processes, and proper documentation controls in place to minimise risks such as contamination, batch inconsistencies, or product defects. During a GMP audit, inspectors typically review several core pharmaceutical operations to assess compliance.
Key areas they evaluate during GMP inspections
Batch Manufacturing Records (BMR/BPR)
Auditors closely review Batch Manufacturing Records (BMR) or Batch Production Records (BPR) to ensure that every stage of manufacturing is properly documented. This includes raw material usage, processing parameters, in-process checks, operator signatures, and final batch approvals by Quality Assurance (QA). Accurate batch documentation is essential for demonstrating product consistency and regulatory traceability.
Quality Control (QC) and Laboratory Processes
Inspectors assess laboratory testing procedures performed by the Quality Control department, including analytical testing of raw materials, intermediates, and finished products. They review test results, analytical methods, stability studies, and compliance with validated testing protocols to ensure the product meets established specifications.
Traceability Across the Supply Chain
GMP regulations require full traceability of materials and products. Auditors verify whether companies can trace active pharmaceutical ingredients (APIs), excipients, intermediates, and finished products across procurement, production, packaging, and distribution stages. This traceability is critical for recalls, pharmacovigilance, and regulatory reporting.
Standard Operating Procedures (SOPs)
Auditors evaluate whether well-defined Standard Operating Procedures (SOPs) exist for manufacturing, quality testing, equipment handling, cleaning procedures, and documentation practices. They also verify whether employees follow these procedures consistently during day-to-day operations.
Equipment Calibration, Qualification, and Maintenance
Pharmaceutical manufacturing relies heavily on specialised equipment such as reactors, mixers, tablet presses, and filling lines. Inspectors review equipment calibration records, preventive maintenance logs, and qualification documentation to confirm that machinery operates accurately and reliably.
Training and Personnel Qualification
Another key area auditors review is employee training records. GMP requires that personnel involved in production, quality assurance, and laboratory operations are properly trained and qualified. Auditors check training logs to ensure employees understand SOPs, GMP requirements, and their specific responsibilities.
Deviation Management and CAPA
During inspections, regulators also review how companies handle deviations, non-conformances, and quality incidents. This includes documentation of deviations, root cause analysis, and implementation of Corrective and Preventive Actions (CAPA) to prevent recurrence.
How Pharma ERP Software in the UK Simplifies GMP Compliance and Audit Readiness
Pharmaceutical manufacturers in the UK are increasingly turning to Pharma Manufacturing Software like BatchMaster ERP to strengthen GMP compliance and simplify audit preparation. It is designed for process manufacturing industries and integrates production, quality control, traceability, and compliance documentation into a single system. This structured approach helps pharmaceutical companies maintain consistent processes, accurate records, and full traceability even during GMP inspections.
Below are some ways BatchMaster ERP supports GMP compliance and helps organisations remain audit-ready:
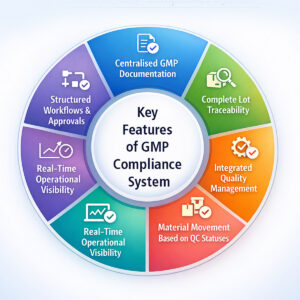
Centralised GMP Documentation
BatchMaster ERP allows pharma companies to maintain GMP-related documents such as Batch Manufacturing Records (BMR), Batch Production Records (BPR), SOPs, quality documents, and compliance reports within one centralised system.
This structured documentation helps organisations maintain regulatory documentation standards aligned with MHRA inspection requirements and EU-GMP documentation guidelines. As a result, records remain properly organised and can be retrieved quickly whenever auditors request them during regulatory inspections.
Complete Lot Traceability
Pharmaceutical ERP systems provides end-to-end traceability, enabling manufacturers to track APIs, excipients, intermediates, and finished products from procurement through production, packaging, and distribution, and also vice versa. This level of traceability supports product recall readiness and pharmacovigilance obligations expected by global regulators such as the EMA. In the event of an audit or product recall, companies can quickly trace product history and material usage.
Integrated Quality Management
BatchMaster Pharma ERP integrates quality processes directly into the manufacturing workflow. It helps manage quality testing, inspection plans, deviation tracking, and quality approvals, ensuring that production activities follow structured quality frameworks such as the ICH Q10 Pharmaceutical Quality System. This helps ensure quality issues are properly recorded, investigated, and resolved.
Structured Workflows and Approvals
BatchMaster ERP Pharma supports structured workflows for activities such as batch processing, quality approvals, and batch release. With system-based approvals and digital audit trails, companies can maintain documentation in line with data integrity principles such as ALCOA+, clearly showing who performed an activity, when it occurred, and whether it was reviewed and approved.
Movement of material based on QC statuses
BatchMaster ERP allows movement of material based on quality statuses. Statuses apart from pass are refrained from moving in the supply chain. Quarantined status’ lots are put on hold till the defined period. QC fail lots are destroyed or returned to vendor as the case, thereby meeting GMP in the easiest of ways.
Reporting
During regulatory inspections, auditors often request detailed reports related to batch production, quality checks, material usage, and traceability. BatchMaster pharmaceutical manufacturing ERP can generate structured reports quickly, helping organisations demonstrate adherence to global pharmaceutical quality expectations defined by the WHO GMP framework.
Real-Time Operational Visibility
With integrated dashboards and reporting tools, BatchMaster ERP manufacturing software UK provides real-time visibility into production processes, batch status, inventory levels, and quality checks. This enables teams to proactively identify risks and maintain consistent compliance with pharmaceutical quality governance and internal audit standards.
So, these are some of the aspects where BatchMaster ERP helps. If your pharmaceutical manufacturing business is looking to simplify GMP compliance and strengthen audit preparedness, BatchMaster ERP can help you achieve a more structured, compliant, and efficient operation.
Request a demo with our experts today to learn how BatchMaster ERP can support your GMP compliance and regulatory audit readiness.





